Hopkins Develops Single Test for 8 Cancers
Researchers at Johns Hopkins Kimmel Cancer Center in Baltimore have developed a single blood test that screens for eight common cancer types and helps identify the location of the cancer. The work could someday lead to routinely screening people and catching tumors before they cause symptoms, when the chance for a cure is best.
Johns Hopkins researchers have developed CancerSEEK, a single test that can detect eight differenct cancers. (Photo: iStock)
As cancer cells die, they shed some of their mutated DNA into the blood. The test, called CancerSEEK, evaluates levels of eight cancer proteins and the presence of cancer gene mutations from DNA in the blood. The test screens for eight common cancers that cause more than 60 percent of cancer deaths in the U.S. — cancers of the colorectum, lung, breast, ovary, liver, stomach, pancreas, esophagus, colorectum, lung and breast. The last five don’t currently have screening tests.
“The use of a combination of selected biomarkers for early detection has the potential to change the way we screen for cancer, and it is based on the same rationale for using combinations of drugs to treat cancers,” said Nickolas Papadopoulos, senior author and a professor of oncology and pathology.
The findings were published online by Science on Jan. 18.
“Circulating tumor DNA mutations can be highly specific markers for cancer. To capitalize on this…we sought to develop a small yet robust panel that could detect at least one mutation,” said Joshua Cohen, a student at the Johns Hopkins University School of Medicine and the paper’s first author. “In fact, keeping the mutation panel small is essential to minimize false-positive results and keep such screening tests affordable.”
The test rarely found cancer that wasn’t there. It had greater than 99 percent success. “Very high specificity was essential because false-positive results can subject patients to unnecessary invasive follow-up tests and procedures to confirm the presence of cancer,” said Kenneth Kinzler, a professor of oncology and co-director of the Ludwig Center. Only seven — less than 1 percent — of 812 healthy patients tested positive.
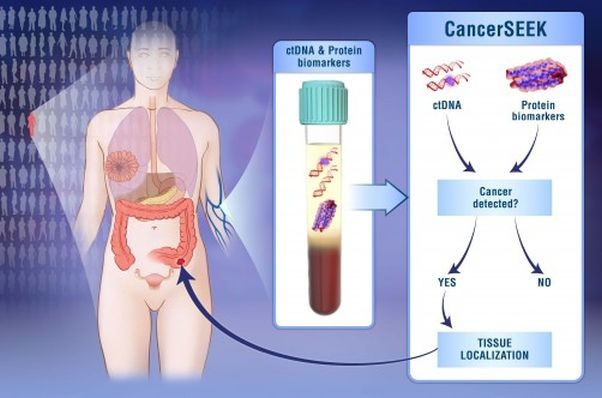
CancerSEEK can detect eight types of cancers and the location. (Graphic: Elizabeth cook and Kaitlin LIndsay/Johns Hopkins)
In blood samples from 1005 patients with the eight types of cancers that had not metastasized yet, the test detected an average of 70 percent of the cases. Success rates ranged from 98 percent for ovarian cancer to 33 percent for breast cancer. For the five cancers that have no screening tests, detection ranged from 69-98 percent.
Although the current test does not detect every cancer, it identifies many that would likely otherwise go undetected. It is also not known if the test will detect cancer in patients without symptoms or with smaller tumors that don’t shed as much DNA. It is also not known how having other inflamatory diseases will affect test results.
“Many of the most promising cancer treatments we have today only benefit a small minority of cancer patients, and we consider them major breakthroughs,” said Dr. Bert Vogelstein, co-director of the Ludwig Center and professor of oncology. “If we are going to make progress in early cancer detection, we have to begin looking at it in a more realistic way, recognizing that no test will detect all cancers. This test represents the next step in changing the focus of cancer research from late-stage disease to early disease, which I believe will be critical to reducing cancer deaths in the long term.”
The investigators said that a test that will be used routinely for cancer screening must have a cost in line with or less than other currently available screening tests for single cancers, such as colonoscopy. They envision that the CancerSEEK test will eventually cost less than $500.
Larger studies of the test are currently under way.
Johns Hopkins Medicine contributed to this report.




